세부 프로그램
GBC1
개회식/기조&특별 강연
기본정보
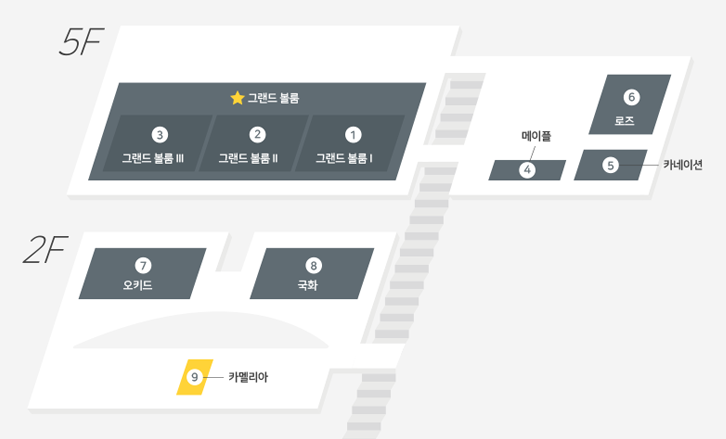
| 일시 | 6.27(수) 13:30~18:00 | 장소 | 그랜드볼룸 5F 안내 |
|---|---|---|---|
| 설명 | 바이오의약품 산업의 현재와 미래 발전 전망을 확인합니다. 신성장동력 바이오경제가 ‘사람 중심 경제’로 나아가기 위한 방향과 비전을 제시합니다. |
||
상세일정
| 시간 | 발표제목 | 발표자 | 자료 | 북마크 | 난이도 |
|---|---|---|---|---|---|
| 13:13~13:20 (7') | 오프닝 퍼포먼스 | - | 즐겨찾기 |
최상
 |
|
| 13:20~13:40 (20') | 개회식 |  |
즐겨찾기 |
상
 |
|
| [세션 1] 기조 연설 | |||||
| 13:40~14:10 (30') | 4차 산업혁명, BT와 IT의 융합을 위하여 |
 마리안젤라 시망오 WHO 사무차장
|
 |
즐겨찾기 |
중
 |
| 13:13~13:20 (7') |
하
 |
||||
| 13:13~13:20 (7') |
최하
 |
||||
[GBC 2] Recombinant Protein Products Forum
Current Trends and Future of Biosimilars
BAT1406 is an adalimumab biosimilar that has been systematically compared to the original drug during the R&D processes, including a pharmacodynamic evaluation in acute and chronic inflammation animal models. Through specific binding to TNF-α and the neutralization effect, BAT1406 blocks interaction of TNF-α with cell surface TNF receptors p55 and p75 and thus inhibits its inflammatory effect.
Adalimumab was the world's first approved fully human anti-TNF-α monoclonal antibody and has been approved for eight different indications including rheumatoid arthritis, psoriatic arthritis, psoriasis, ankylosing spondylitis, Crohn's disease, ulcerative colitis, hidradenitis suppurativa, and uveitis.
Adalimumab was the world's first approved fully human anti-TNF-α monoclonal antibody and has been approved for eight different indications including rheumatoid arthritis, psoriatic arthritis, psoriasis, ankylosing spondylitis, Crohn's disease, ulcerative colitis, hidradenitis suppurativa, and uveitis.
 발표자료.pdf
발표자료.pdf
난이도 : 중